Parts of your RV can be susceptible to various types of corrosion, including galvanic.
By Steve Froese, F276276
January 2024
This month I will discuss a rather advanced topic that can have negative impacts on recreational vehicles: galvanic corrosion. This electro-chemical reaction can result in a “frustrating at best, damaging at worst” situation with your RV.
Galvanic corrosion, also called dissimilar metal corrosion or bimetallic corrosion (it is not electrolysis), refers to damage caused when two or more dissimilar metals make contact in the presence of an electrolyte. When a galvanic couple forms, one of the metals becomes the anode (think of electrons flowing toward the metal) and corrodes faster than it would by itself. The other becomes the cathode (electrons flowing away from the metal) and corrodes more slowly than it would alone. We can think of the anode as being more reactive and the cathode as being less reactive in the electrolyte. Water is an electrolyte, so, simply put, galvanic corrosion can occur whenever two or more dissimilar metals contact each other in the presence of water.
Either metal in the couple may or may not be prone to corrosion by itself. However, when contact with a dissimilar metal occurs, the self-corrosion rates change. Corrosion of the anode accelerates while corrosion of the cathode decelerates, or even stops.
The driving force for galvanic corrosion is the potential voltage difference between the materials in the couple. This was discovered in the late part of the 18th century by Luigi Galvani during a series of experiments with the exposed muscles and nerves of a frog. Galvani found that the muscles and nerves contracted when connected to a bimetallic conductor. The principle was later put into a practical application by Alessandro Volta, who built the first electrical cell in 1800. Bearing only a slight resemblance to the batteries of today, his version consisted of two types of metal disks, separated by cardboard disks soaked with acid or salt solution. This is the basis of all modern wet-cell batteries.
A famous case of galvanic corrosion is that of the Statue of Liberty. The skin of the statue is composed of copper, whereas the internal frame is made of wrought iron. The designer and builder of the statue were aware of galvanic corrosion, which is why shellac-coated asbestos was used as an insulating layer between the two metals. However, in the 1980s, it was discovered that the shellac had deteriorated, leaving areas where the two metals came into contact with each other. This resulted in the iron superstructure rusting because of moisture in the air. The original insulating layer was replaced with Teflon, and, along with further restorative work, the statue was refurbished.
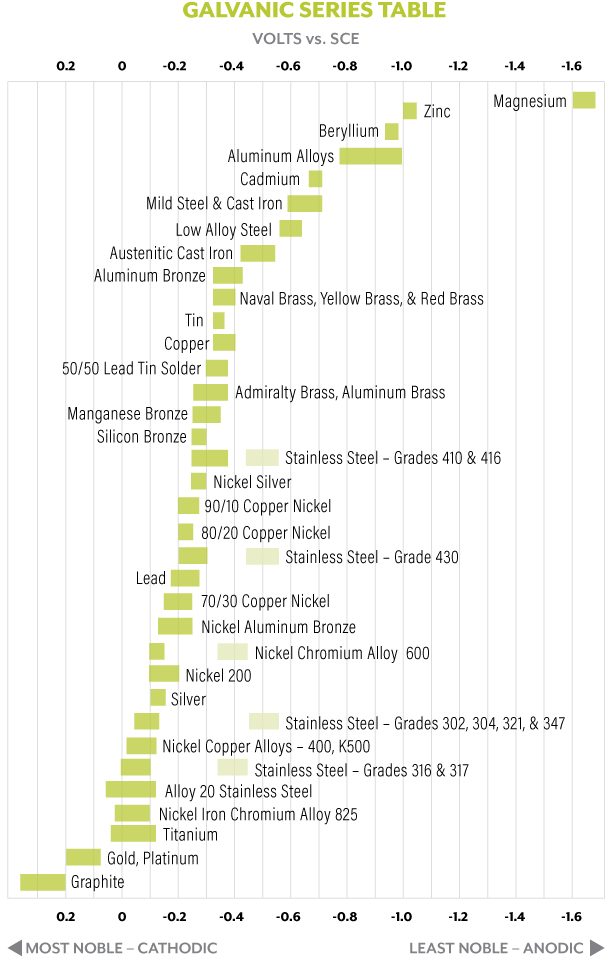
Noble metals are those that are resistant to corrosion and have outstanding resistance to oxidation. In a bimetallic couple, the less noble material (the material with less resistance to corrosion) will become the anode and tend to corrode at an accelerated rate compared with the corrosion tendency of the metal by itself. The more noble material will act as the cathode. The nobility of a metal can be determined by referring to the galvanic series table. This table lists the relative nobility of a metal when exposed to seawater, as measured in volts. Galvanic corrosion can be one of the most common forms of corrosion, as well as one of the most destructive.
So, how do we read the galvanic series table and what does this have to do with your RV? We won’t go into detail regarding this chart, but the importance is in the voltage value of the metals in question. We need to determine the relative voltage between the pair of metals by subtracting the individual values. For example, the primary metals used in RVs are steel and aluminum (although we could find zinc, nickel, etc.). To prevent galvanic corrosion, the relative volt-age must be less than 0.15 volts. Note that in the absence of an electrolyte or contact between the two metals, corrosion cannot occur.
Let’s examine the case of steel versus aluminum. The voltage for aluminum averages about -0.9, whereas the steel voltage is around -0.7. The difference between these two metals is 0.2 volts, higher than the recommended 0.15. This is just an average, but it does indicate the two metals are clearly susceptible to galvanic corrosion. In this example, galvanic corrosion is likely to occur in the presence of water and when the metals are contacting each other. A perfect example of this is when a plastic plug in an aluminum water heater tank drain is replaced with a steel one instead. In this case, we have a relative voltage of about 0.2; the metals are in contact with each other; and water is most definitely present. Teflon tape would not be effective in preventing contact across the entire mating surface. This is why using a steel (or other type of metal) plug in an aluminum hot-water tank often results in galvanic corrosion severe enough to “weld” the plug to the tank.
In the case of a steel hot water tank, such as a Suburban, galvanic corrosion principles are intentionally utilized to prevent the steel tank from corroding. In this case, an anode rod, generally made from aluminum or magnesium, is exposed to the water in the tank. Since the anode rod is made of a less noble metal than the hot-water tank itself, the rod is sacrificed to protect the steel tank. The anode rod can be easily replaced as it wears down from the galvanic corrosion. If you have one of these water heaters in your RV, you can observe this chemical reaction yourself.
Remember that one of the requirements for corrosion to occur is the presence of an electrolyte. In this case, it’s the water. It is not necessary to consider bimetallic pairings in completely dry areas, such as in RV construction; however, in the case of water intrusion, galvanic corrosion does occur. For instance, consider the roof molding on an RV with a rubber roof. The rubber is stretched down the sidewall of the RV, and the perimeter moldings are screwed to the sidewall. Also, the rubber is stapled to the sidewall before the moldings are installed. Since many RVs are now constructed using aluminum framing, we have a situation where galvanic corrosion can occur. The staples are likely to be steel or zinc, as are the screws. These fasteners are secured through the aluminum moldings and RV framing. As long as the walls stay dry, this is not an issue. However, if water enters the sidewall, generally through damage or lack of seal maintenance, both water damage and corrosion can result. This is especially true in the case of prolonged exposure. Also, the smaller the contacting surface area of the bimetallic couple, the less desirable the condition, since the galvanic current is concentrated over a smaller area. Rapid thickness loss of the dissolving anode tends to occur under these conditions. So, in the case of staples, screws, and nails, a total loss of integrity can result as the fastener completely corrodes and disintegrates.
Other areas of an RV can experience corrosion as well, especially in wet environments, so please take the time and effort to thoroughly inspect and maintain your RV, particularly when it comes to seal inspection. Look for other areas on your RV where galvanic corrosion may occur, such as frame and rail components where dissimilar metals come into contact with each other.
Send your troubleshooting questions to Steve Froese at techtalk@fmca.com. The volume of correspondence may preclude personal replies. Not all responses will apply in every instance. Some situations may require a visual inspection and hands-on testing. If you choose to follow any procedures outlined in this column, first satisfy yourself that neither personal nor product safety will be jeopardized. If you feel uncomfortable about a procedure, stop and make an appointment with an RV service facility.
You may also want to consult the FMCA Forums (https://community.fmca.com) to see whether your question has already been addressed or, if not, to post it.
Recalls
Looking for the latest RV-related recall information? Visit FMCA.com for a list of the most recent recalls issued by RV and component manufacturers. Or, to search for recalls, investigations, and complaints by vehicle year, make, model, and VIN, visit www.nhtsa.gov/search-safety-issues. NHTSA’s Vehicle Safety Hotline is (888) 327-4236.