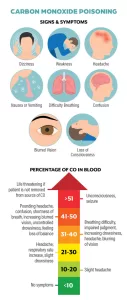
This colorless, odorless, and tasteless gas can be deadly and is present in fumes produced by engines, grills, furnaces, water heaters, and more.
By Steve Froese, F276276
January 2024
The deaths of three U.S. Marine Corps members from carbon monoxide (CO) poisoning in North Carolina this past July is a stark reminder of the dangers of toxic gases. This is especially relevant for RV users, since we could potentially be exposed to CO produced by damaged or maladjusted appliances.
The armed forces members died in a parked car while sitting at a gas station. According to various media reports, discussion resulted afterward regarding how this may have occurred. In a vehicle, malfunctions usually can be seen and/or heard. A common cause of CO leaking into the interior of a car is a damaged exhaust system. This is generally accompanied by excessive and/or unrecognized noise. I have been in a car with a damaged exhaust pipe, and it is unbearable after more than a few miles.
 Older vehicles, such as the one the Marines reportedly were in, can suffer from corrosion and rusting engine and body components. This may result in the release of toxic combustion gases into the engine compartment or directly into the cabin through holes in the floor. In the former case, the lethal gas can get sucked into the intake vents that lead to the interior cabin. As mentioned, it is unusual for this to happen without accompanying noise. While CO itself is odorless and colorless, other gases present in a vehicle exhaust system are not, so it is likely the Marines heard and smelled something prior to the tragic event. This makes their deaths even more mysterious.
Older vehicles, such as the one the Marines reportedly were in, can suffer from corrosion and rusting engine and body components. This may result in the release of toxic combustion gases into the engine compartment or directly into the cabin through holes in the floor. In the former case, the lethal gas can get sucked into the intake vents that lead to the interior cabin. As mentioned, it is unusual for this to happen without accompanying noise. While CO itself is odorless and colorless, other gases present in a vehicle exhaust system are not, so it is likely the Marines heard and smelled something prior to the tragic event. This makes their deaths even more mysterious.
This article is not meant to be a chemistry lesson, but I believe a very good way to describe carbon monoxide and how it forms is by explaining the chemistry in a basic sense. I will try to keep things in layperson’s terms and attempt to avoid making it sound like a college lecture. So, this article contains basic chemistry principles related to the combustion process. Those who’d like a slightly deeper dive should read the sidebar titled “The Process Of Combustion.”
Carbon monoxide is created as a byproduct of incomplete combustion. In other words, it’s a combustion reaction not going the way it is supposed to. Carbon monoxide is highly toxic, and since it has no smell and is invisible, it is easy to find yourself overcome once you are exposed. While vehicle exhaust system leaks are likely to be noticeable due to a sound or smell from other components or chemical compounds, propane-burning RV appliances are not likely to emit any warning sounds or smells, other than possibly a roaring flame.
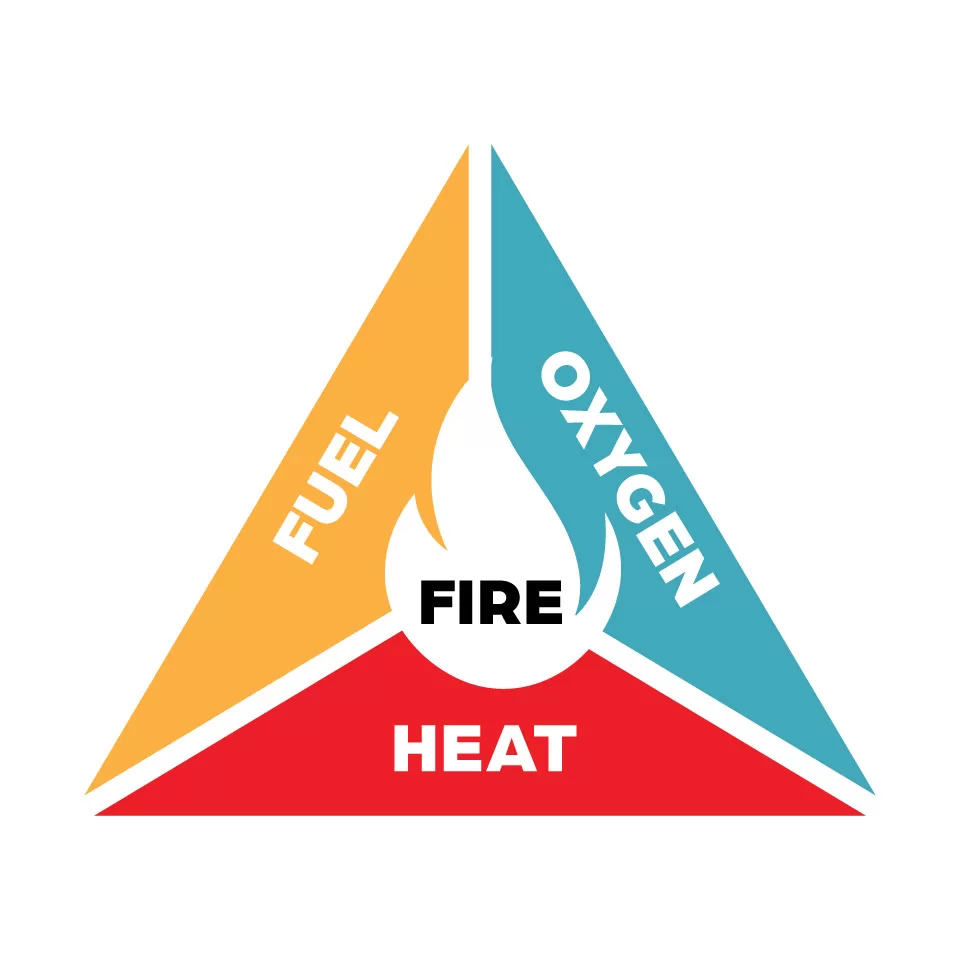
RVs that utilize onboard propane systems have several appliances that burn propane. Any of these could emit CO if damaged or improperly adjusted. Let’s briefly discuss the chemical reaction involved in combustion. The accompanying “fire triangle” diagram illustrates the combustion reaction graphically.
To achieve the flame (energy) in the middle of the triangle, three inputs are necessary. These required elements are oxygen (or another oxidizer), a fuel source (such as propane), and heat (this would be an ignitor, lighter, or match). When combined in the correct ratios, the result is heat and light in the form of a flame. The flame will be sustained as long as the three inputs are provided. Remove any of the inputs, and the flame will extinguish.
What is not shown on the triangle are the byproducts of combustion, which are water (in the form of steam) and carbon dioxide (CO2). To be more specific, these are the byproducts of complete combustion. Complete combustion occurs when the oxygen-to-fuel mixture is set to the correct ratio. RV and other combustion appliances are designed to allow a specific amount of air (oxygen) into the reaction. On some appliances, the ratio can be adjusted within a narrow range to alter the flame characteristics.
If an appliance is damaged or incorrectly adjusted, the result can be incomplete combustion. This is potentially a very dangerous scenario in which one of the byproducts is not carbon dioxide but rather carbon monoxide. CO forms when the oxygen concentration is reduced in a combustion reaction. Two main causes of incomplete combustion are too much heat or too little oxygen.
Higher flame temperatures result in CO, since the reaction occurs faster and consumes oxygen at a higher rate. So, if any propane- burning appliances are damaged or misadjusted and burn too hot or don’t introduce enough oxygen into the combustion process, a likely byproduct is carbon monoxide. As evidenced by the tragedy of the Marines, this often results in a fatal outcome.
If CO is present in our system, our body’s hemoglobin (the oxygen-transport protein present in red blood cells) combines with it and diminishes the hemoglobin’s oxygen-carrying capacity, which is a fatal condition. Because CO is colorless and odorless in the absence of other gases, CO can easily kill you without you ever knowing it.

It is important to have a working carbon monoxide detector on board your RV.
Since you wouldn’t be able to see or smell the dangerous CO gas, you must rely on a functional carbon monoxide alarm in your RV to keep you and your family safe. Be sure to regularly test all your alarms, which should consist of at least one each of smoke, propane, and CO. If your alarms are battery operated, I recommend replacing the batteries every six months. If your geographical area observes daylight saving time, it is helpful to use the act of changing your clocks as a reminder to replace the batteries in these devices. Also pay attention to the manufacture date stamped on the alarms themselves and the manufacturer’s instructions. They generally should be replaced every seven to 10 years, or in the event of a failure. Modern CO alarms are required to have end-of-life warning indicators, so be sure to pay attention to any flashing and/or beeping from your detector. This is important, since the alarms lose their ability to effectively detect the toxic gas as they age.
Another important way to help prevent your gas appliances from creating CO in your RV is to have your propane system inspected by a professional RV service technician at least annually. The technician will check your system for proper operating pressure, functionality, leaks, and damage. I can’t stress enough the importance of regular propane system maintenance and CO detector testing and replacement, as it could spare you the same fate as the poor Marines.
As a final comment, while propane is not toxic, the vapor will replace the oxygen in the air in a confined space, resulting in possible suffocation. And, of course, propane is highly flammable. Propane is a very safe and efficient fuel if used properly and treated with respect, but never expose yourself to a confined space while propane is present. An example would be using a propane appliance indoors.
Propane safety is of paramount importance to RV owners, so please take the time and make the effort to keep the propane system properly maintained and resist the temptation to ignore or delay required appliance repairs.
THE PROCESS OF COMBUSTION
Exothermic combustion reactions, or those that create energy, must involve hydrocarbons or any compound consisting of hydrogen and carbon atoms, such as propane.
Let’s consider a combustion reaction involving propane. The chemical formula for propane is C3H8, or three carbon atoms and eight hydrogen atoms per molecule. Other hydrocarbons include butane, methane, and natural gas. The “g” in the equation below refers to the vapor state; all the reactants and products of combustion are in vapor (gaseous) form.
C3H8(g)+5O2(g)→3CO2(g)+4H2O(g) + energy (heat)
The chemical equation must be properly balanced on each side to achieve the desired result of carbon dioxide and water vapor.
A balanced equation, also referred to as “complete combustion,” requires that the number and type of atoms be the same on each side of the equation. This necessitates a precise amount of oxygen on the reactant (left) side of the equation, which contributes its atoms to both the carbon dioxide and water vapor on the product side. For the equation above, we can mathematically determine the equation is balanced. On the reactant side of the equation, we have a total of 10 oxygen atoms (5O2). This must be maintained on the right side, which
we can see as six oxygen molecules converted to carbon dioxide, plus four oxygen molecules converted to water vapor. While the oxygen has converted from pure oxygen on the reactant side to contribute both to the CO2 and H2O products, the number of total oxygen atoms remains equal on both sides of the equation.
If the amount of oxygen in a combustion reaction is too low, CO is the likely result. This is referred to as “incomplete combustion” and can result from too much heat or too little oxygen. Let’s examine the equation above with half the oxygen required:
2C3H8(g)+7O2(g)→6CO(g)+8H2O(g) + energy (heat)
In this case, we have CO as a product instead of CO2. Applying the same math as above, we can see we have 14 oxygen atoms on the left and 14 on the right. However, the difference is that one of the products is carbon monoxide instead of carbon dioxide.